Highlights:
- Rheumatoid arthritis is a long-term autoimmune disorder.
- The mechanism of this condition targeting the joints’ cells.
- Excess expression of IL-6 can contribute in the disease.
- Tocilizumab (TCZ) is the first therapy used.
- Vitamin D(Vit D) has a crucial role in inhibiting the expression of IL-6 as reported by recent studies.
Abstract
Rheumatoid arthritis (RA), an autoimmune disease leads to various cardiovascular, pulmonary, physiological disorders. It results due to the interplay of environmental and genetic factors. Il-6 is a cytokine which proliferates and differentiates B-cells. Although they contribute to the host-defence system, their persistent expression may cause immune diseases. IL-6 is a major factor causing rheumatoid arthritis. It initiates the signalling cascade which activates the cell proliferation. The first therapy which was discovered against RA by targeting IL-6 is Tocilizumab (TCZ) which is a monoclonal antibody and prevents binding of IL-6 from its trans membrane receptors. Though several therapies have been discovered with high success rate, the intensive research is required which can help to affect more specific targets.
Keywords: autoimmune disease, interleukins, B-cell, T-cell
Rheumatoid Arthritis
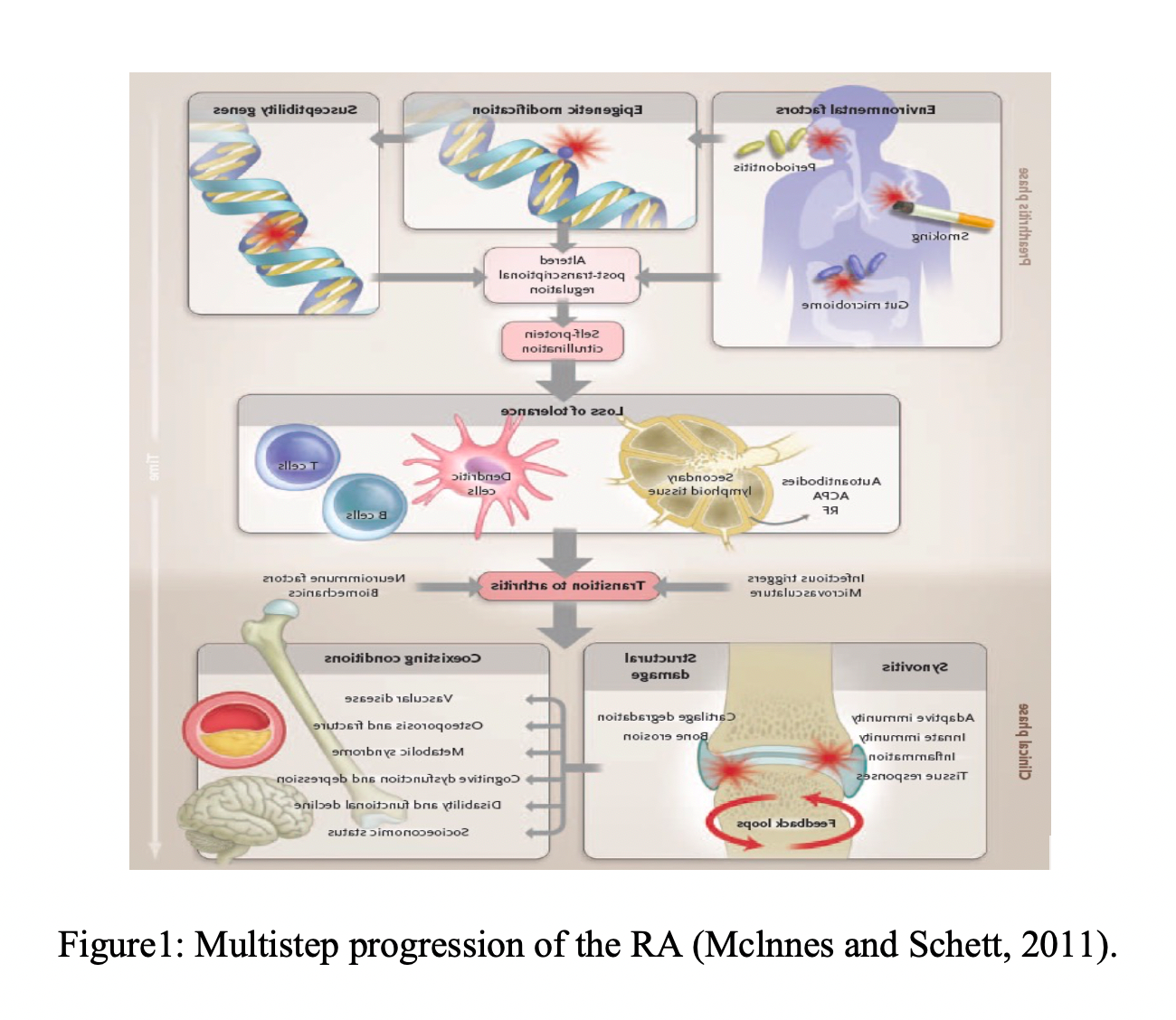
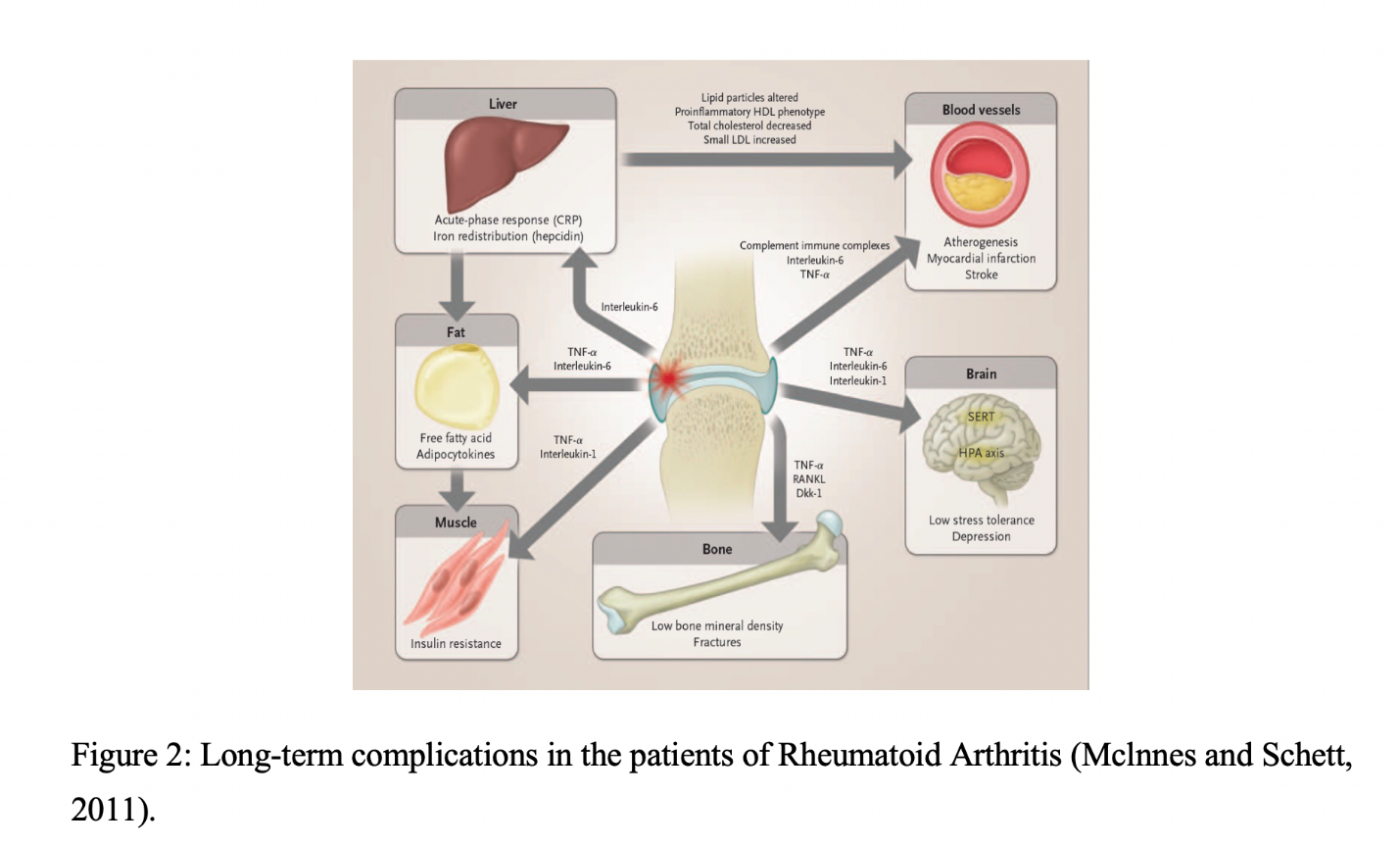
Rheumatoid Arthritis is a very common autoimmune disease which is characterized by synovial inflammation, production of rheumatoid factor (RF) which further causes the cardiovascular, pulmonary, physiological and skeletal disorders (Mclnnes and Schett,2011) Figure 1. Long term complications in patients have been described in Figure 2. RA is the outcome of various environmental and genetic factors. Though more exact cause of RA needs to be explored, recent findings suggest that 80% of RA patients carry epitope of the HLA-DRB1*04 cluster (Smolen et al., 2007). Single Nucleotide Polymorphism genotyping across MHC has identified some additional alleles that also cause the risk of RA including A1-B8-DR3(Choy, 2012).
Pathogenesis of Rheumatoid Arthritis
The pathogenesis of RA has been widely studied by researchers. With the help of genome-wide 1methods as well as other conventional molecular analysis, the genetic construct of this disease has been very well understood (Catrina et al., 2016). In one of the reviews it was estimated that the risk and progression of this disease is controlled by around 100 loci (Okada et al., 2014). The very important loci are that of the genes that code for HLADR01/04, a typical MHC class II molecule which is important in the recognition of auto-reactive peptides by T cells and other co-stimulatory pathways and thus affecting CD40, CD28, cytokine and chemokine receptors. Other influencers include enzymes responsible for post translational modification like PADI that catalyses the modification of peptidylarginine to citrulline, pathways that regulate intracellular functioning like STAT 3 and PTPN22(Ytterberg et al., 2015; Vassallo et al., 2014). The above-mentioned factors all challenge the immune responses by either activating the immune cells like IgM and IgG or failing their regulation pathways (Konig et al., 2016).
Initiation of the disease is known as pre-rheumatoid arthritis stage may then be characterized by production of autoantibodies that detects a wide spectrum of proteins like carbamylated and acetylated peptides, post transnationally modified ones with citrulline residue. Local microtrauma, activation of local complement system or any slight micro-vascular injury may lead to articular localization of the autoantibodies. Synovitis may be promoted by the local release of tumor necrosis factor α and interleukin 8 associated with circulation of autoantibodies that may identify the citrullinated proteins and immune complexes leading to bone damage (Krishnamurty et al., 2015; Pfeifle et al., 2017). The synovial lesion that has once been initiated will encounter a large inflow of migrating B cells, plasma cells, T cells, macrophages, mast cells and activated stromal cells like synovial fibroblasts. The propagation of the disease may be seen when these cells interact with the immune mediators like cytokines and chemokines (Pfeifle et al., 2017).
Interleukins
Interleukins are the cytokines which was first expressed by leucocytes and later on it was found to produce from different body cells. They play essential role in activation of the immune cell, their proliferation, differentiation, migration, adhesion, etc. They exhibit both anti-and pro-inflammatory properties. Following are the few other properties of cytokines/interleukins:
- They are produce in response to pathogens or other antigens which produce inflammatory response.
- mRNA encoding interleukins are unstable and once they are synthesised they are secreted immediately.
- IL can both up and down regulate the mechanism either by induction or by encoding the inhibitory IL receptors or cause the antibody switching of isotypes of B-cell, differentiation of T-cells in TH1 and TH2.
The following table (Table 1) describe some different classes of interleukin and their roles:
| Class | Functions | Reference |
| IL-1 | Secreted by macrophages, B-cells, astrocytes. It causes lymphocyte activation and also apoptosis in many cell types. | Bachmann and Oxenius, 2007 |
| IL-2 | T cells produce IL-2. It causes proliferation and activation of NK cells. | Gabunia and Autieri, 2015 |
| IL-3 | Produced by T-cells. It functions as a multilineage colony-stimulating factor. | Lin et al., 2015; Shabgah et al., 2017 |
| IL-4 | CD4+T cells (Th2) synthesize IL-4. It act as B-cell growth factor and causes IgE and IgG1 isotype selection | Gabunia and Autieri, 2015 |
| IL-8 | Monocytes and fibroblasts secretes IL-8. It causes neutrophil chemotaxis, angiogenesis, superoxide release. | Guerra et al., 2017 |
| IL-6 | T and B lymphocytes, fibroblasts and macrophages secretes IL-6. It differentiate B-cells | Persaud et al., 2016 |
| IL-17 | Produced by Th-17. It helps in the secretion of Il-6. | Wang et al.,2016; Takada et al., 2017. |
| IL-27 | T cell secretes it which activates STAT-1 and STAT-3 and also secretes IL-10. | Persaud et al., 2016 |
| IL-21 | NK cells and CD4+ T lymphocytes secrete IL-21. It proliferates NK cells. | Belghith et al., 2018 |
Interleukin 6 (IL-6) and it signalling
IL-6 is a glycoprotein with pleiotropic activity. Earlier it was discovered as the B-cell differentiation factor (BCDF) which is derived from T-cells and use for the proliferation of B-cells (Yoshizaki et al., 1982). IL-6 triggers the signalling by the binding to an 80 kDa transmembrane IL-6 receptor (IL-6R). After binding to IL-6R the complex with IL-6 and IL-6R transduce the signalling molecule gp130 which further activates the downstream signalling of Janus kinase (JAK) (Stahl et al., 1994). This is the classical signalling pathway. The IL-6R is expressed on only limited cells such as hepatocytes and some leukocytes. JAK is a member of tyrosine kinase family and its phosphorylation activate the transcription of STAT 3 and hyperphosphorylation of mitogen activated protein kinases (MAPKs). STAT 3 further stimulates the expression of several genes leading to the induction of cell growth and differentiation (Stahl et al., 1995). The schematic presentation of signalling is explained in Figure3.
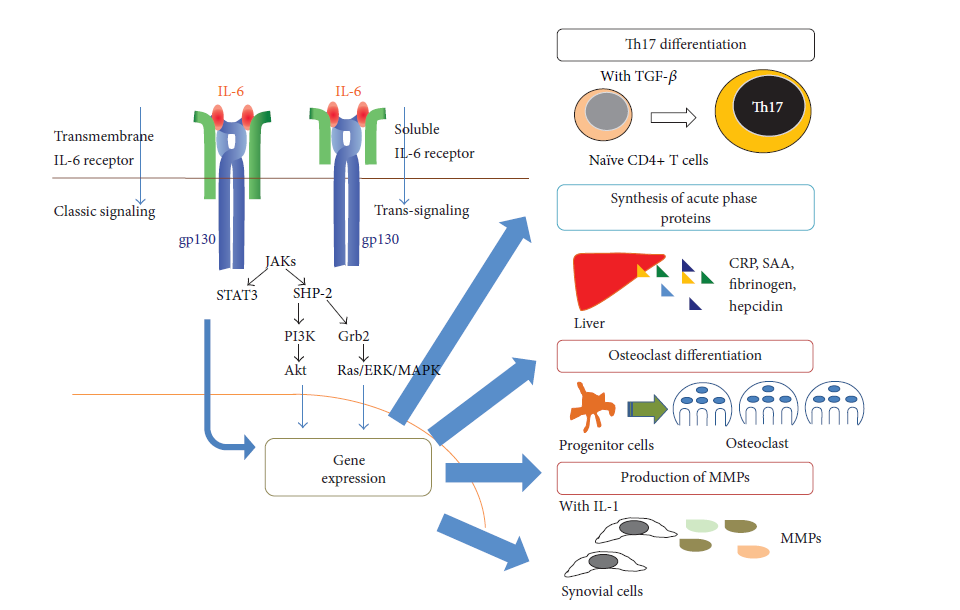
Figure 3:
Interleukin-6 cell signalling mechanism
Role of IL-6 in Rheumatoid Arthritis
A normal synthesis of IL-6 promotes the host-defence mechanism. However, uncontrolled persistent of IL-6 can leads to the origin of several immune mediated diseases. RA is one of the immune disease which occurs due to the abnormality of IL-6 (Yoshida and Tanaka, 2014). In addition to IL-6, many cytokines have also played important role in the pathogenesis of RA. Earlier it was considered that IL-6 derived from the helper T-cells which activates the B-cells but recent findings have suggested that they also act as the regulator of CD4+T cells and causing their activation and differentiation. Besides activating JAK-STAT signalling cascade, IL-6 influences T-cell into differentiate in TH2 cell by upregulating nuclear factors of activated T-cells (NFAT)c2 and c-maf and inhibiting IFNγ signalling (Rincon et al., 1998). Moreover, presence of TGF-β promotes IL-6 to differentiate T-cell in TH17 by upregulating STAT3 mediated retinoid orphan receptor (ROR)γt and inhibits TGF-β induced regulatory T-cells (Treg) differentiation (Korn et al., 2009; Kimura et al., 2010) which promotes predominance of Th17 over Treg in the effector CD4+ T cells which is the major cause for the development of RA and several immune mediated diseases.
IL-6 targeted therapies for RA treatment:
The above literature regarding pathology of RA has led to the concept that IL-6 targeting therapy may be the strategy to cure RA. Tocilizumab (TCZ), a humanized anti-IL-6 monoclonal antibody of the IgG1 class was developed which blocks the IL-6 mediated signalling cascade (Sato et al., 1993). It is considering as first line biologics for the treatment of active RA. TCZ works through two mechanisms either binding to the IL-6 and preventing it to bind to receptor or by dissolving the receptors.
Recent studies indicated that Vit D, a steroid hormone is well known for its role in the calcium metabolism and bone homeostasis (Rosen et al., 2012), plays the important role in curing RA by inhibiting the expression of IL-6 (Gopal et al., 2019). However, it seems that Vitamin D and its analogy interact with the immune system and causes T cell proliferation and further inhibit the expression of pro‐inflammatory cytokines such as interleukin (IL)‐6, IL‐17 and IL‐23 which are involved in RA pathogenesis (van Hanburg et al., 2012; Ranganathan et al., 2013). Vitamin D upregulates the osteoprotegrin (OPG)/RANK L ratio mediating anti-inflammatory response and also decreasing IL-6 levels (Feng et al., 2013).
Conclusions and perspectives
In light of the above knowledge, it can be concluded that though immune system is there to protect our body from antigens, their over expression may lead cause harm to our body. It is significant to study the signalling of IL-6 more intensively due to its involvement in triggering onset of RA. Despite intensive studies providing the sufficient therapies for curing RA, there is still a lacuna which needs to be filled. Use of TCZ is the first line to use against RA and showed improvements in systemic inflammation, it is also necessary to study whether these antibodies are efficient in treating the fundamental abnormalities of RA. Vitamin D is known to be efficient for treating RA but more detailed action of mechanism need to be explored.
References
- immunoregulation and back again. EMBO reports, 8(12), 1142-1148.
- Belghith, M., Bahrini, K., Kchaou, M., Maghrebi, O., Belal, S., & Barbouche, M. R. (2018). Cerebrospinal fluid IL-10 as an early stage discriminative marker between multiple sclerosis and neuro-Behçet disease. Cytokine, 108, 160-167.
- Catrina, A. I., Joshua, V., Klareskog, L., & Malmström, V. (2016). Mechanisms involved in triggering rheumatoid arthritis. Immunological reviews, 269(1), 162-174.
- Choy, E. (2012). Understanding the dynamics: pathways involved in the pathogenesis of rheumatoid arthritis. Rheumatology, 51(suppl_5), v3-v11.
- Feng, X., Lv, C., Wang, F., Gan, K., Zhang, M., & Tan, W. (2013). Modulatory effect of 1, 25-dihydroxyvitamin D 3 on IL1β-induced RANKL, OPG, TNFα, and IL-6 expression in human rheumatoid synoviocyte MH7A. Clinical and Developmental Immunology, 2013.
- Gabunia, K., & Autieri, M. V. (2015). Interleukin-19 can enhance angiogenesis by Macrophage Polarization. Macrophage, 2(1), e562.
- Gopal, K., Thevarajah, M., Ng, C. M., & Raja, J. (2019). Effects of vitamin D on disease activity and serum interleukin‐6 in rheumatoid arthritis. International journal of rheumatic diseases.
- Guerra, E. S., Lee, C. K., Specht, C. A., Yadav, B., Huang, H., Akalin, A., … & Levitz, S. M. (2017). Central role of IL-23 and IL-17 producing eosinophils as immunomodulatory effector cells in acute pulmonary aspergillosis and allergic asthma. PLoS pathogens, 13(1), e1006175.
- Kimura, D., Miyakoda, M., Kimura, K., Honma, K., Hara, H., Yoshida, H., & Yui, K. (2016). Interleukin-27-producing CD4+ T cells regulate protective immunity during malaria parasite infection. Immunity, 44(3), 672-682.
- Konig, M. F., Abusleme, L., Reinholdt, J., Palmer, R. J., Teles, R. P., Sampson, K., … & Moutsopoulos, N. M. (2016). Aggregatibacter actinomycetemcomitans–induced hypercitrullination links periodontal infection to autoimmunity in rheumatoid arthritis. Science translational medicine, 8(369), 369ra176-369ra176.
- Korn, T., Bettelli, E., Oukka, M., & Kuchroo, V. K. (2009). IL-17 and Th17 Cells. Annual review of immunology, 27, 485-517.
- Krishnamurthy, A., Joshua, V., Hensvold, A. H., Jin, T., Sun, M., Vivar, N., … & Magnusson, M. (2015). Identification of a novel chemokine-dependent molecular mechanism underlying rheumatoid arthritis-associated autoantibody-mediated bone loss. Annals of the rheumatic diseases, annrheumdis-2015.
- Lin, P. Y., Jen, H. Y., Chiang, B. L., Sheu, F., & Chuang, Y. H. (2015). Interleukin‐21 suppresses the differentiation and functions of T helper 2 cells. Immunology, 144(4), 668-676.
- McInnes, I. B., & Schett, G. (2011). The pathogenesis of rheumatoid arthritis. New England Journal of Medicine, 365(23), 2205-2219.
- Okada, Y., Wu, D., Trynka, G., Raj, T., Terao, C., Ikari, K., … & Graham, R. R. (2014). Genetics of rheumatoid arthritis contributes to biology and drug discovery. Nature, 506(7488), 376.
- Pfeifle, R., Rothe, T., Ipseiz, N., Scherer, H. U., Culemann, S., Harre, U., … & Haugg, B. (2017). Regulation of autoantibody activity by the IL-23–T H 17 axis determines the onset of autoimmune disease. Nature immunology, 18(1), 104.
- Persaud, L., De Jesus, D., Brannigan, O., Richiez-Paredes, M., Huaman, J., Alvarado, G., … & Sauane, M. (2016). Mechanism of action and applications of interleukin 24 in immunotherapy. International journal of molecular sciences, 17(6), 869.
- Ranganathan, P., Khalatbari, S., Yalavarthi, S., Marder, W., Brook, R., & Kaplan, M. J. (2013). Vitamin D deficiency, interleukin 17, and vascular function in rheumatoid arthritis. The Journal of rheumatology, 40(9), 1529-1534.
- Rincón, M., Enslen, H., Raingeaud, J., Recht, M., Zapton, T., Su, M. S. S., … & Flavell, R. A. (1998). Interferon‐γ expression by Th1 effector T cells mediated by the p38 MAP kinase signaling pathway. The EMBO journal, 17(10), 2817-2829.
- Rosen, C. J., Adams, J. S., Bikle, D. D., Black, D. M., Demay, M. B., Manson, J. E., … & Kovacs, C. S. (2012). The nonskeletal effects of vitamin D: an Endocrine Society scientific statement. Endocrine reviews, 33(3), 456-492.
- Sato, K., Tsuchiya, M., Saldanha, J., Koishihara, Y., Ohsugi, Y., Kishimoto, T., & Bendig, M. M. (1993). Reshaping a human antibody to inhibit the interleukin 6-dependent tumor cell growth. Cancer Research, 53(4), 851-856.
- Smolen, J. S., Aletaha, D., Koeller, M., Weisman, M. H., & Emery, P. (2007). New therapies for treatment of rheumatoid arthritis. The Lancet, 370(9602), 1861-1874.
- Stahl, N., & Yancopoulos, G. D. (1994). The tripartite CNTF receptor complex: activation and signaling involves components shared with other cytokines. Journal of neurobiology, 25(11), 1454-1466.
- Stahl, N., Farruggella, T. J., Boulton, T. G., Zhong, Z., Darnell, J. E., & Yancopoulos, G. D. (1995). Choice of STATs and other substrates specified by modular tyrosine-based motifs in cytokine receptors. Science, 267(5202), 1349-1353.
- van Hamburg, J. P., Asmawidjaja, P. S., Davelaar, N., Mus, A. M., Cornelissen, F., van Leeuwen, J. P., … & Lubberts, E. (2012). TNF blockade requires 1, 25 (OH) 2D3 to control human Th17-mediated synovial inflammation. Annals of the rheumatic diseases, 71(4), 606-612.
.Vassallo, R., Luckey, D., Behrens, M., Madden, B., Luthra, H., David, C., & Taneja, V. (2014). Cellular and humoral immunity in arthritis are profoundly influenced by the interaction between cigarette smoke effects and host HLA-DR and DQ genes. Clinical Immunology, 152(1-2), 25-35.
.Wang, X., Wei, Y., Xiao, H., Liu, X., Zhang, Y., Han, G., … & Li, Y. (2016). A novel IL‐23p19/Ebi3 (IL‐39) cytokine mediates inflammation in lupus‐like mice. European journal of immunology, 46(6), 1343-1350.
.Yoshida, Y., & Tanaka, T. (2014). Interleukin 6 and rheumatoid arthritis. BioMed research international, 2014.
.Ytterberg, A. J., Joshua, V., Reynisdottir, G., Tarasova, N. K., Rutishauser, D., Ossipova, E., … & Malmström, V. (2015). Shared immunological targets in the lungs and joints of patients with rheumatoid arthritis: identification and validation. Annals of the rheumatic diseases, 74(9), 1772-1777.
.Yoshizaki, K., Seo, Y. O. S. H. I. T. E. R. U., Nishikawa, H. I. R. O. Y. A. S. U., & Morimoto, T. A. K. E. T. O. S. H. I. (1982). Application of pulsed-gradient 31P NMR on frog muscle to measure the diffusion rates of phosphorus compounds in cells. Biophysical journal, 38(2), 209-211.
